|
These electrons enter the electron transport chain of the mitochondria via reduction. IUBMB Life published by Wiley Periodicals LLC on behalf of International Union of Biochemistry and Molecular Biology. The malate-aspartate shuttle (sometimes simply the malate shuttle) is a biochemical system for translocating electrons produced during glycolysis across the semipermeable inner membrane of the mitochondrion for oxidative phosphorylation in eukaryotes. MAS NADH/NAD ratio aspartate citrate-malate cycle glycerol-1-P cycle inborn errors reductive carboxylation. This illustrates the vitality of ongoing MAS research. The year 2019 saw the discovery of two new inborn errors in the MAS, deficiencies in malate dehydrogenase 1 and in aspartate transaminase 2 (GOT2). 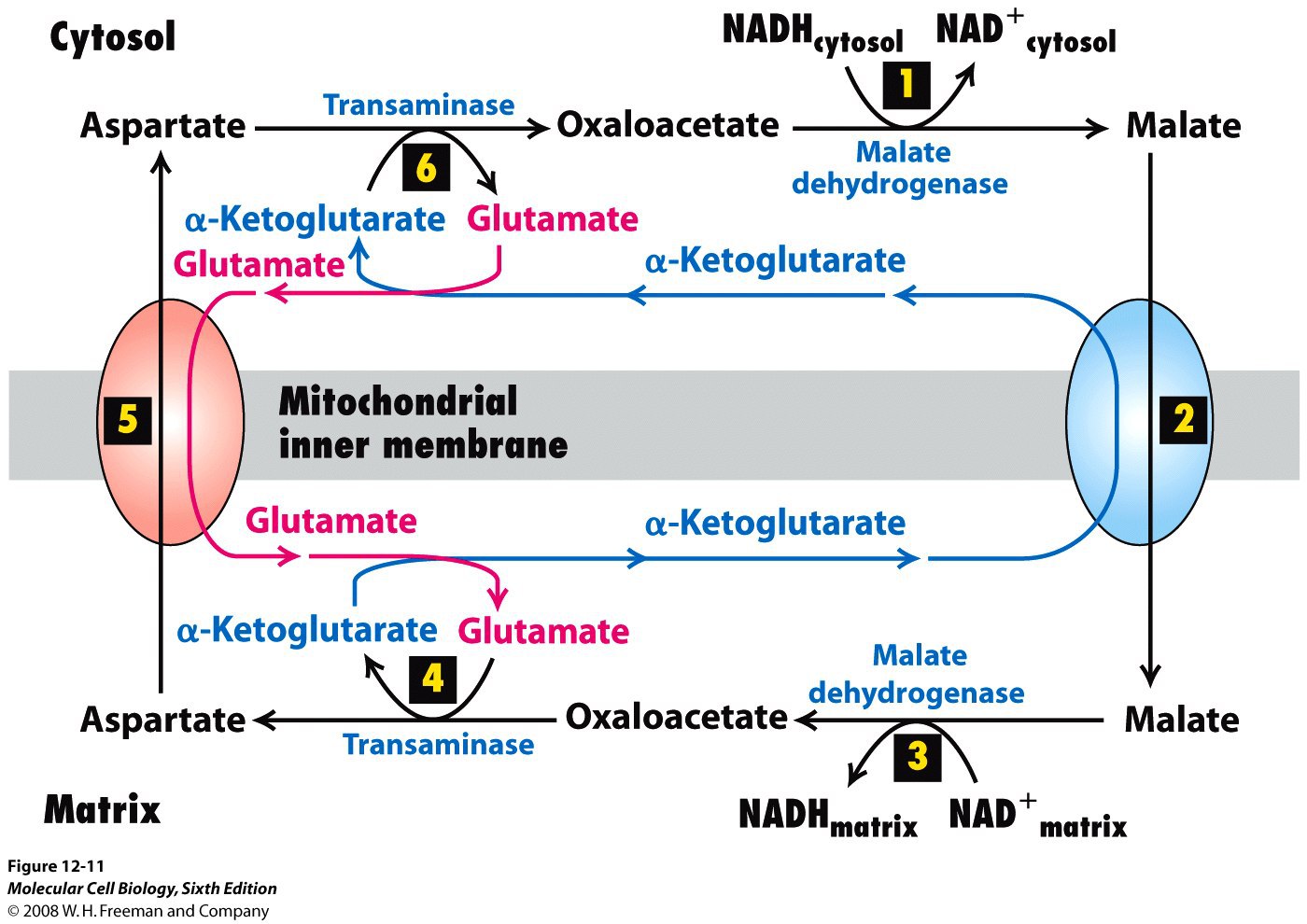
Most recently, the focus has been on the role of the MAS in tumors, on cells with defects in mitochondria and on inborn errors in the MAS. The MAS is still a very active field of research. This makes the MAS in practice uni-directional toward oxidation of cytosolic NADH, and explains why the free NADH/NAD ratio is much higher in the mitochondria than in the cytosol. Only in the 1970s, LaNoue and coworkers discovered that the efflux of aspartate from mitochondria, an essential step in the MAS, is dependent on the proton-motive force generated by the respiratory chain: for every aspartate effluxed, mitochondria take up one glutamate and one proton. The MAS was soon adopted in the field as a major pathway for NADH oxidation in mammalian tissues, such as liver and heart, even though the energetics of the MAS remained a mystery. The MAS was initially proposed as a route for the oxidation of cytosolic NADH by the mitochondria in Ehrlich ascites cell tumor lacking other routes, and to explain the need for a mitochondrial aspartate aminotransferase (glutamate oxaloacetate transaminase 2 ). The malate-aspartate shuttle (sometimes simply the malate shuttle) is a biochemical system for translocating electrons produced during glycolysis across the semipermeable inner membrane of the mitochondrion for oxidative phosphorylation in eukaryotes. The malateaspartate NADH shuttle components are novel metabolic longevity regulators required for calorie restriction-mediated life span extension in yeast. OxPhos colon cancer l-lactate mLDH malate-aspartate shuttle.This article presents a personal and critical review of the history of the malate-aspartate shuttle (MAS), starting in 1962 and ending in 2020. The elevated glycolysis in cancer cells is proposed to be one of the mechanisms acquired to accelerate oxidative phosphorylation. We demonstrated that the malate-aspartate shuttle exerts control over NAD + /NADH homeostasis to maintain activity of mitochondrial lactate dehydrogenase and to enable aerobic oxidation of glycolytic l-lactate in mitochondria. We investigated the role of mitochondria-associated malate-aspartate and lactate shuttles in colon cancer cells as potential regulators that couple aerobic glycolysis and oxidative phosphorylation. We hypothesized that activities of glycolysis and oxidative phosphorylation are coordinated to maintain redox compartmentalization. However, there are a growing number of studies that show that mitochondria remain highly oxidative under glycolytic conditions. Elevated glycolysis underlying the Warburg effect is an established signature of cancer metabolism. 
Therefore, many cancer types, including colon cancer, reprogram mitochondria-dependent processes to fulfill their elevated energy demands. Within the context of these competing drives aerobic glycolysis is inefficient for the cancer cellular energy economy. Metabolism in cancer cells is rewired to generate sufficient energy equivalents and anabolic precursors to support high proliferative activity.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
